FDA recalls heart device again after patients die.
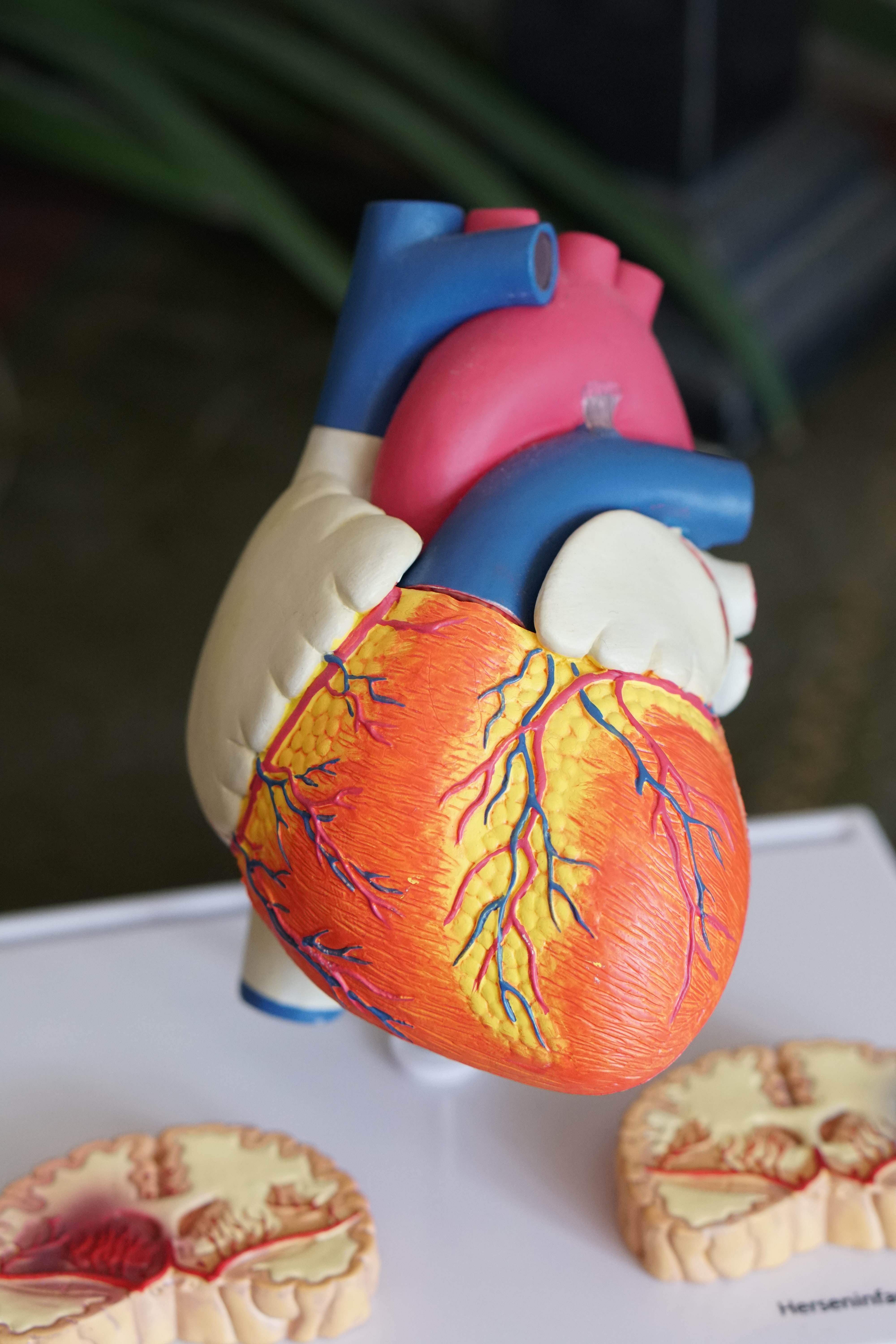
Medtronic’s HeartWare HVAD ventricular assist device has been once again recalled by the Food and Drug Administration (FDA) after the agency learned it included a design flaw that has led to patient deaths. The HeartWare HVAD ventricular assist device is used to help keep blood flowing during the final stage of heart failure. According to the company’s website, A VAD consists of: “A pump that is attached to a ventricle inside the body; An external controller, which is a small computer that monitors the pump; A driveline cable, which connects the pump to the controller; Power sources that run the pump and controller.”
The device is implanted in a patient’s chest, in a sac known as the pericardial space. The HVAD Pump is connected directly to the heart, where it draws oxygen-rich blood. The driveline, or cable is fed through an abdominal incision and is connected to the pump.
Medtronic has included “Important Safety Information” regarding the device on its site, including the statement, “This device may cause serious and life- threatening events, including stroke.” The site goes on to warn consumers, “There are many known risks associated with the surgical procedure as well as after the HVAD has been implanted. These risks include, but are not limited to, death, stroke, device malfunction, blood clots, bleeding, other types of heart failure, infection, red blood cell destruction, and total body infection.”
The FDA said the device “has issues that can cause the outflow graft to tear and the strain relief screw to break before the heart pump is implanted.” The agency said the issue “may cause serious patient harm, including dizziness, loss of consciousness, bleeding, fluid buildup around the heart, additional medical procedures and death.”
According to the warning, “Medtronic has received 92 complaints related the pre-implant pump assembly process, which includes both the strain relief screw breaking and outflow graft tears.” Four deaths have also been reported. At least twelve class 1 recalls have been issued by the FDA on the HeartWare device since 2014, which is the most serious category and means the device can be fatal.
Medtronic didn’t pull the devices from the market after the recall but advised users to follow additional steps in its instructions for use. The notice included further instructions. The agency advised consumers and others affected to:
“Review the steps listed…for assembly and attachment of the outflow graft and strain relief for future HVAD System implants.;
Closely inspect the graft after assembly and before implantation for any possible tears.
Continue to practice standard peri-operative and immediate post-operative patient management to detect for this issue.;
Complete the enclosed Physician Confirmation Form and either return it to your Field Representative or email to RS.CFQFCA@medtronic.com to indicate you received the notification.”
“Medtronic is working with regulatory agencies for approval on a design change that is intended to reduce the risk of damage during assembly of the outflow graft and strain relief and attachment onto the pump,” a company spokesperson responded. “Medtronic will provide training on this design change after the necessary regulatory approvals are obtained.”
Sources:
Medtronic heart pump recalled again after link to 4 patient deaths

Join the conversation!