A Dallas judge’s ruling resulted in a pyrrhic victory for Johnson & Johnson.
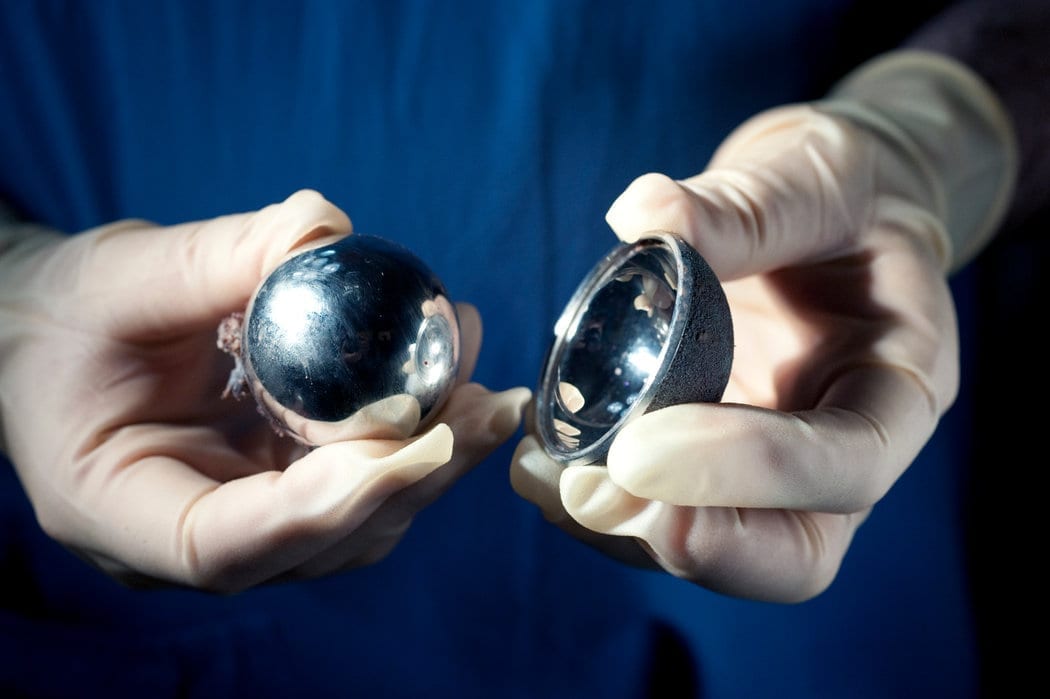
The latest and possibly last chapter in a long-lasting court case came to a close at the beginning of January. For two years, Johnson & Johnson had been fighting six plaintiffs’ accusations of negligence. At the forefront of the drama was a simple medical device. Pinnacle hip implants, which act as a socket-like connection between the femur and pelvis, were sold to customers suffering from localized arthritis pain and past fractures. The implants – a DePuy product – had certain shortcomings which the lawsuit claims were covered up.
In December, a federal jury ordered Johnson & Johnson to pay over one-billion dollars in compensatory and punitive damages. The payout was slashed nearly in half at the beginning of the New Year.
US District Judge Ed Kinkeade was quoted in the Bloomberg Press as saying, “Constitutional considerations limit the amount a plaintiff may recover in punitive damages.” The national Supreme Court had set the limit for punitive damages as being no greater than ten times the plaintiffs’ actual damages, which figured out at about $35 million.
While the defense may have won a victory which scaled the scope of their financial obligation from “colossal” to “massive,” they have by no means sailed out of rough water. Thousands of lawsuits relating to Pinnacle are pending in courts across the United States. To date, Johnson & Johnson and DePuy have paid out over $2.5 billion to people adversely affected by faulty hip-replacement systems.
Findings from the early Pinnacle and DePuy ASR CL Acetabular System trials in 2010 noted how extensive the injuries and risks posed by the device could be. Both DePuy and J&J were found at fault for not having disclosed product problems to physicians and patients alike. When a recall was ordered, preliminary findings suggested a staggering rate of device failure within a five-year period. Doctors in the United States and United Kingdom gave figures ranging between 13 and 49% – an astonishing number which ran counter to corporate claims that Pinnacle and Acetabular were the long-lasting replacements for ceramic and plastic hip implants.
The consequences of Pinnacle breaking down or wearing out were not minor instances of discomfort. Metal components contained within Acetabular and Pinnacle sometimes led to infection, bone fractures, and severe pain. Shards torn off and tossed about the body by friction or collisions could wander through way through the pelvis, lodging in bony structures and occasionally causing sicknesses as severe as necrosis, which is cell death associated with gangrene and tissue decomposition.

Most of the lawsuits brought against the two companies – including the slashed $1.2 billion payout – brought into the legal limelight dangerous defects as well as attempts to cover up damages from disaffected or distraught customers. Johnson & Johnson, so far as the law is concerned, engaged in a pattern of near-malicious attempts to delay product recalls as well as deceptive marketing practices. With more cases heading to the courtroom, the defense is not unlikely to be paying out further damages.
Sources:
Johnson & Johnson hit with over $1 billion verdict on hip implants
Johnson & Johnson Just Got Hit With a $1B Verdict Over Faulty Hip Implants
J&J Judge Slashes $1 Billion Verdict Over Pinnacle Hip Implants
J&J Hip Failure Rate as High as 49 Percent, U.K. Doctors Say

Join the conversation!