The legal system provides a critical pathway to hold powerful corporations accountable.
Beyond the fine print on a medication label lies a stark reality: pharmaceutical products can cause profound harm. Each year in the United States, adverse drug reactions are responsible for over 1.5 million emergency department visits, and nearly 500,000 of those incidents are serious enough to require hospitalization.
Consumers place immense trust in medications approved by the Food and Drug Administration (FDA), but this approval does not grant manufacturers immunity when their products cause unforeseen injury. This guide demystifies the complex world of product liability law, explaining the legal grounds consumers have to hold pharmaceutical companies accountable, the types of claims they can make, and the typical process for seeking justice and compensation.
Understanding Product Liability in the Pharmaceutical Industry
At the heart of every dangerous drug case are core legal principles designed to protect consumers. These principles hold that the companies profiting from a medication are also responsible for the harm it may cause. Navigating this legal landscape requires a clear understanding of a manufacturer’s duties and the limits of regulatory oversight, which are often not as absolute as the public perceives.
What is Product Liability?
Product liability is the legal responsibility a manufacturer or seller holds for producing or selling a faulty product that causes injury. In the pharmaceutical industry, this means drug companies have a legal duty to ensure their medications are reasonably safe for their intended use.
This critical duty is not a single checkpoint but an ongoing obligation that extends from a drug’s initial design and chemical formulation through its manufacturing process and all the way to its final marketing and labeling. Any failure along this chain can expose a company to legal action if a patient is harmed as a result.
The Myth of Absolute FDA Protection
A common misconception is that FDA approval provides a bulletproof shield against lawsuits. While manufacturers frequently use this approval as a central part of their defense, courts have consistently affirmed that regulatory clearance does not protect a company from liability if its warnings are later proven inadequate or misleading.
The FDA sets minimum safety standards for a drug to enter the market, but the manufacturer retains an ongoing duty to monitor its performance, collect data on adverse events, and update its warnings as new risks emerge. The fact that hundreds of drugs are recalled by the FDA each year demonstrates that serious issues are frequently discovered long after a product has been approved and distributed to the public.
The Critical Role of Causation
The central challenge in any dangerous drug lawsuit is proving “causation”—that is, establishing a direct and provable link between the medication and the patient’s injury. This is far more complex than simply showing that a person took a drug and then became ill.
Proving causation requires sophisticated medical evidence, testimony from scientific and medical experts who can explain the drug’s mechanism of action, and a meticulous review of the patient’s medical history to rule out other potential causes. This complexity is precisely why specialized legal representation, such as a dangerous drugs lawyer, is essential to build a compelling case against a well-funded pharmaceutical corporation.
The Three Pillars of a Dangerous Drug Case
When a patient is harmed by a medication, their legal team will typically build a case around one or more specific types of claims. These claims are not mutually exclusive but represent distinct arguments about where the manufacturer failed in its duty to ensure patient safety.
- Defective Design: A defective design claim alleges that the drug is inherently unsafe, meaning its risks outweigh its potential benefits, regardless of how perfectly it was manufactured or how clearly it was labeled.
- Manufacturing Defects: This claim focuses on a flaw that occurred during production, packaging, or shipping—such as contamination or incorrect dosage. In the first half of 2024 alone, 384.2 million units of pharmaceutical products were recalled in the U.S.
- Failure to Warn (Marketing Defects): This is the most common claim. It contends that the manufacturer failed to provide adequate warnings about side effects. A recent case study involves GLP-1 drugs, where over 4,400 plaintiffs allege manufacturers failed to warn about stomach paralysis (gastroparesis).
Common “Failure to Warn” violations include:
- Actively downplaying the severity of side effects in marketing.
- Omitting critical risk information from the official label.
- Failing to update warnings after new dangers emerge in post-market surveillance.
- Promoting the drug for unapproved “off-label” uses without specific warnings.
Understanding Mass Torts and MDLs
When a single drug injures thousands of people, individual lawsuits are often consolidated into a Mass Tort, usually managed as a Multi-District Litigation (MDL). This streamlines the process by grouping similar cases before one judge to handle evidence exchange (discovery) efficiently.
The General Timeline of a Mass Tort Lawsuit
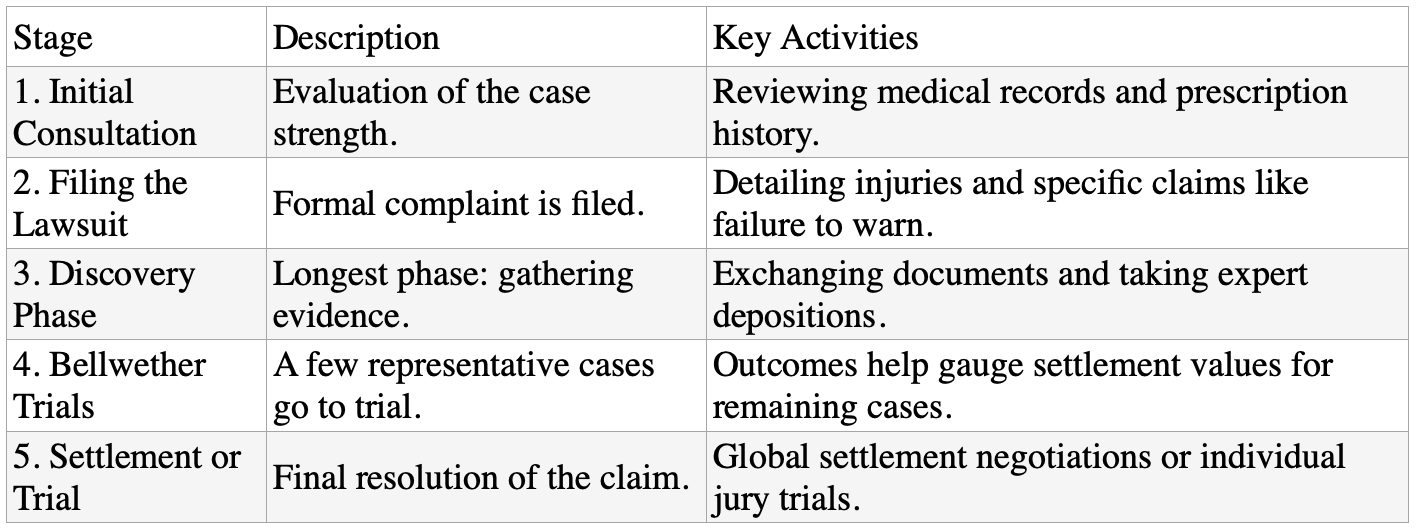
A System Designed for Accountability
The legal system provides a critical pathway to hold powerful corporations accountable. With median jury awards in product liability cases nearing $4 million in recent years and massive global settlements like the $26 billion opioid agreement, the system has the power to force change. If you or a loved one has suffered an unexpected injury, understanding your legal rights is the first step toward personal recovery and broader public safety.

Join the conversation!