Swedish hospitals have already stopped using hydroxychloroquine to treat coronavirus, citing its potentially deadly side effects.
President Trump has continued to pressure federal officials into making anti-malaria medications available for coronavirus patients.
According to Reuters, Trump has repeatedly and continuously endorsed the efficacy of two drugs—chloroquine and hydroxychloroquine—as effective counters to novel coronavirus. That is despite a dearth of scientific evidence suggesting that either medication is sufficient to combat the infection’s spread or symptoms.
In fact, Trump’s opinion seems based solely off speculation. While there is limited evidence to suggest that hydroxychloroquine may suppress some coronavirus symptoms, its efficacy has neither been extensively tested nor proven.
Yet the Center for Disease Control—in what Reuters calls a “highly unusual move”—recently published guidance informing physicians across the country that they may be prescribe hydroxychloroquine as a treatment for coronavirus. The CDC’s dosing recommendations diverge from ordinary procedure and may be influenced by “unattributed anecdotes.”
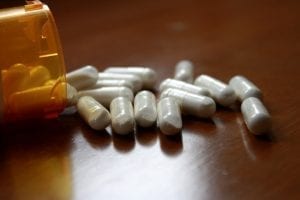
Hydroxychloroquine has a host of potentially lethal side effects, especially when taken by people who are demographically most at-risk for developing serious coronavirus symptoms—the elderly, and those with pre-existing health conditions. The anti-malarial may, for instance, cause cardiac problems. And reports from Sweden, which recently restricted hydroxychloroquine’s use for COVID-19 cases, suggests many patients developed medical problems worse than coronavirus had caused.
“There were reports of suspected more serious side effects than we first thought,” said Professor Magnus Gisslén, chief physician at Sahlgrenska University Hospital infection clinic. “We cannot rule out serious side effects, especially from the heart, and it is a hard-dosed drug. In addition, we have no strong evidence that chloroquine has an effect on COVID-19.”
U.S experts have similarly stressed that using chloroquine and hydroxychloroquine for coronavirus may lead to unpredictable outcomes.
Jeffrey Flier, former dean of Harvard Medical School, said Trump’s enthusiasm for an untested drug poses a fundamental challenge to the United States’ oversight structures.
“The president is short-circuiting the process with his gut feelings,” Flier said. “We are in an emergency and we need to rely on our government to ensure that all these potential therapies are tested in the most effective and objective way.”
However, the Trump administration, as well as its supporters, say “short-circuiting the process” is what needs to be done in a time of medical calamity.
“The President’s top priority is the health and safety of the American people which is why he brought together the federal government and private sector, including doctors, scientists, and medical researchers, for an unprecedented collaboration to expedite vaccine development,” the White House said in a statement to Reuters.
A former government official told the outlet that, in ordinary times, the CDC would never issue guidance encouraging physicians to try unproven treatments.
“In a perfection situation you would never do this,” said the official, who Reuters referred to as a “public health specialist” who recently left a government position. “But if you know what the safety downside is, and the patient is ready to try it, it’s worth a try.”
Sources
Exclusive: Pressed by Trump, U.S. pushed unproven coronavirus treatment guidance


Join the conversation!